Clinicians struggling with screen and alert fatigue from increasing amounts of electronic information say the overload of clinical and patient data has grown into a pernicious problem.
In fact, the very information most crucial to bedside care can become obscured by the sheer volume of data-points, graphs and reports populating the system.
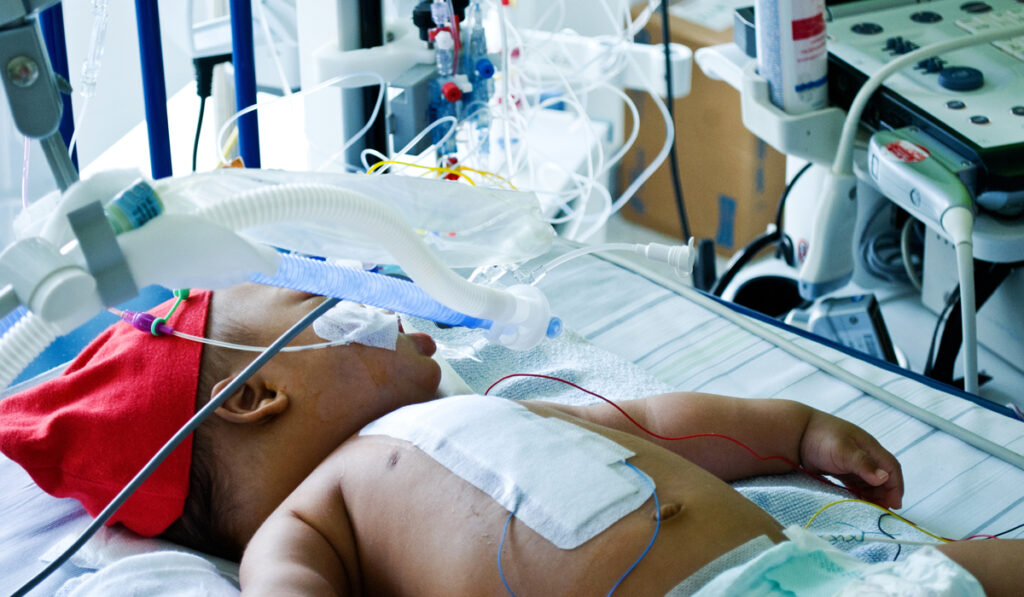
At Monroe Carell Jr. Children’s Hospital at Vanderbilt, physician-researchers are now working on a translational initiative intended to clear the deck of extraneous or second-tier information and bring crucial data forward to the clinician’s attention. Nowhere is the need for such discernment greater than with the fragile patients with single ventricle heart disease.
At the April 2022 Pediatric Academic Societies meeting, Children’s Hospital cardiologist Jeffrey Weiner, M.D., presented his team’s initiative to make crucial echocardiographic information more quickly actionable on behalf of babies with hypoplastic left heart syndrome (HLHS). Weiner converted summary-field information in echocardiogram reports into a dynamic guide for clinicians, incorporating charts, graphs, and other data.
He and his team then defined five highly actionable data points, or descriptors, that paint a picture of an individual baby’s status.
“Cardiologists use echocardiography, in addition to physical exams, as a mainstay for care. The reports are often the key to finding out if there is a canary in the coal mine that suggests that these kids may not be doing as well as they look outwardly,” Weiner said.
Three-Step Palliation
Babies with hypoplastic left heart syndrome (HLHS) are born with the left ventricle missing or severely underdeveloped. In their first week of life, most will undergo a Norwood procedure that enables the right ventricle to perform the tasks of both ventricles. This creates a neo-aorta that sustains the body using a mix of oxygenated blood from the lungs and deoxygenated systemic blood. The surgeon will also create a source of pulmonary blood flow, using artificial material to create a shunt between either the heart or the aorta and the pulmonary arteries.
A second palliative procedure, the Glenn procedure, is performed about four to six months after recovery from the Norwood procedure. It redirects blood flow from the upper body to the lungs and relieves some of the work from the right ventricle. The Fontan procedure, performed at about 3 to 5 years of age, is the definitive surgery, rerouting the blood flow from the lower body to the lungs by connecting the inferior vena cava to the pulmonary artery.
Existential Challenges
Greater use of the Norwood procedure has fueled an increase in the in-hospital survival rate for these babies, whose odds a quarter century ago were extremely low. Today, that rate is approximately 63-74 percent. Still, these children face multiple existential challenges.
Despite improvements in HLHS repair, children continue to have compromised postoperative systemic cardiac output, reduced systemic oxygen delivery, high systemic oxygen extraction, and anaerobic end-organ dysfunction. They also have more surgeries, cardiac catheterizations, and hospitalizations compared with children who have other complex congenital heart lesions.
“In that four-to-six months, between the first and second stage, these babies have a very tenuous existence, because their one ventricle is doing twice the work it was designed to do,” Weiner said. “Failure to thrive is a continual threat because they burn way more calories just to live. Their heart function can go down, they can have leaky valves, or problems with their reconstructed aorta. If they develop blood clots in the heart, all of a sudden they don’t have any pulmonary blood flow, and that can be rapidly fatal.”
Interventions Can’t Wait
Children with HLHS also have a high risk for neurodevelopmental disabilities. While this is associated with syndromic etiology in about 25% of cases, typical oxygen saturation rates hovering around 80 percent “on a good day” underscore the need for vigilance to mitigate neurocognitive impact, Weiner said.
“We asked ourselves what information would enable us, within our sphere of influence as interventionalists, imaging specialists and surgeons, to push toward better outcomes,” he said. “Then, if we are explicit about these actionable items on the echocardiogram report, we can move these babies away from grave danger to intervention earlier.”
Weiner and his team of echocardiography attendings and sonographers identified five areas to highlight that signify patients’ top health needs: functioning status of the interatrial septum, tricuspid valve and great artery; right ventricle functionality; and case summary statements from previous caregivers.
One Playbook
Weiner says this change, adopted at Children’s Hospital in November 2020, enables all caregivers to follow the critical stats from the same playbook and take action swiftly when circumstances require.
“These are data that would prompt us to bring in our interventional cath team if an issue arises with the great arteries or the interatrial septum,” Weiner said. “If the tricuspid valve is leaky, we could alert a surgeon to repair. If their systolic function is deficient, we can ask the child’s intensivist or the cardiologist to change their medication. The key is not just to look for abnormal, but actionable findings, and recognize these needs as close to ‘real-time’ as we can.”