Neuroscientists at Vanderbilt University Medical Center have discovered that a distinct area in the brain contributes to poor prognoses for patients with glioblastoma. The niche contains certain types of unusual cells and, when in contact with a glioblastoma, appears to accelerate tumor growth.
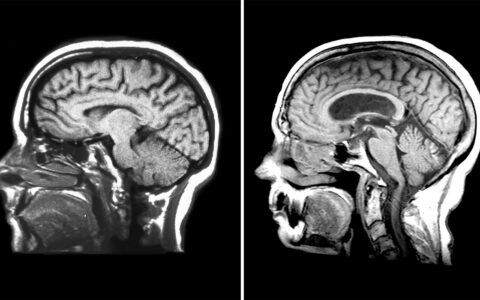
“This niche – the ventricular-subventricular zone – is right next to structures that are highly visible on MRI,” said Rebecca Ihrie, Ph.D., an associate professor of cell and developmental biology and neurological surgery at Vanderbilt. “This has inspired us to pursue an MRI-based diagnostic tool that would give surgical teams insights into biological features of glioblastomas to drive targeted therapies.”
Multifaceted Approach
At the core of the work is a three-way interplay between the cancer, stem and immune cells found in the niche.
Jonathan Irish, Ph.D., an associate professor of cell and developmental biology, and pathology, microbiology and immunology, described research underway at Vanderbilt to tease apart the mechanisms.
“We’ve used machine learning to identify the different types of cancer cells present in tumors that correlate with survival,” Irish said.
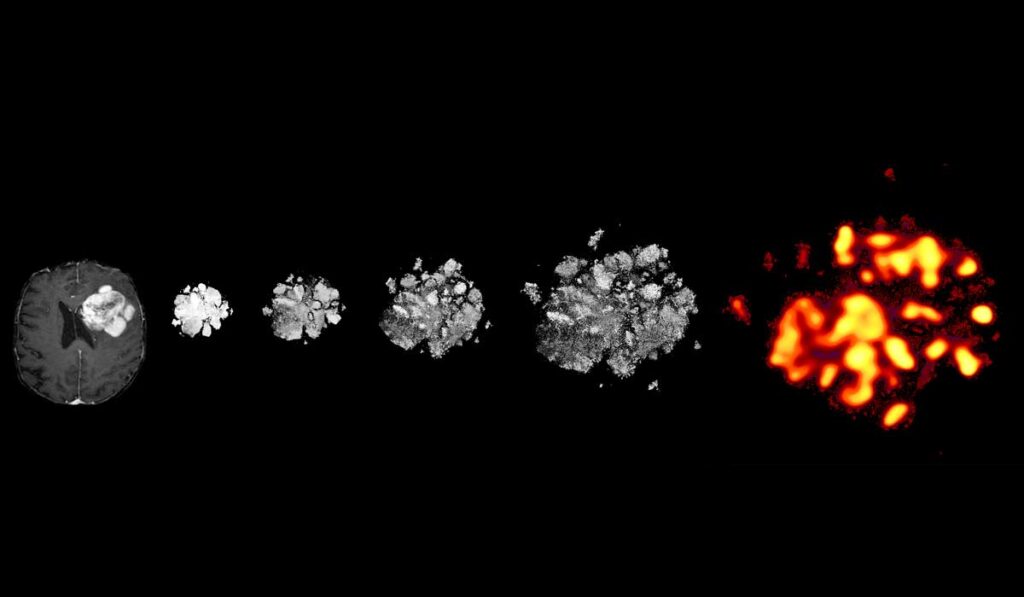
A dataset of two million glioblastoma cells led to Risk Assessment Population IDentification (RAPID) maps that can now be used to identify tumor cells associated with rapid progression and poor survival.
Collaborations with Vanderbilt neurosurgeons are also driving ongoing studies about the location of glioblastomas in the brain, and how physical contact with one stem cell niche and not another drives tumorigenesis.
Finally, the researchers are drilling down on immune cells only found in tumors with niche contact. Preliminary data are helping to define which cells might suppress versus promote glioblastoma growth.
“We believe this is all related. It’s helping us understand the cellular basis of tumors, so we can next link that to MRI,” Irish said.
Cells Tell the Story
With support from the Vanderbilt-Ingram Cancer Center and others, the researchers are now translating their cellular findings into clinical applications.
“With a single non-invasive MRI, we can begin to understand what’s going on with these cancers at the cellular level.”
First, the team takes tumor tissue directly from the operating room and parses out the various cell populations present.
“This has revealed really unusual cell types,” Irish said. “They express proteins that normally do not coexist – neural stem cell proteins and mature neuronal proteins coexisting in one cell, for example.”
Based on these proteins, the researchers can study where the cells came from and where they are most likely to appear. “The cell biology tells the story,” Irish said. “Now in tumor resection, we know the different cell types, and we collect at least three distinct regions for analysis rather than a single mass,” Ihrie explained.
The results may someday help neurosurgeons and neurologists match patients to therapies that target the cellular features observed.
Translating to MRI
So far, the researchers have used immunohistochemical staining and studies of public MRI and genomic data to validate their single-cell analyses.
“We’re beginning to see cellular neighborhoods, or social networks, within the tumor microenvironment,” Irish said.
The findings could provide context for clinical MRI. Identifying the precise location of a glioblastoma on MRI could help predict the types and features of cells present and drive therapy choices.
“With a single non-invasive MRI, we can begin to understand what’s going on with these cancers at the cellular level. That could allow us to design clinical trials around immunotherapies that target particular cell populations, for example,” Irish said.
The researchers already have launched experiments to screen existing medications for their ability to target distinct cells within tissue samples donated by people with glioblastoma.