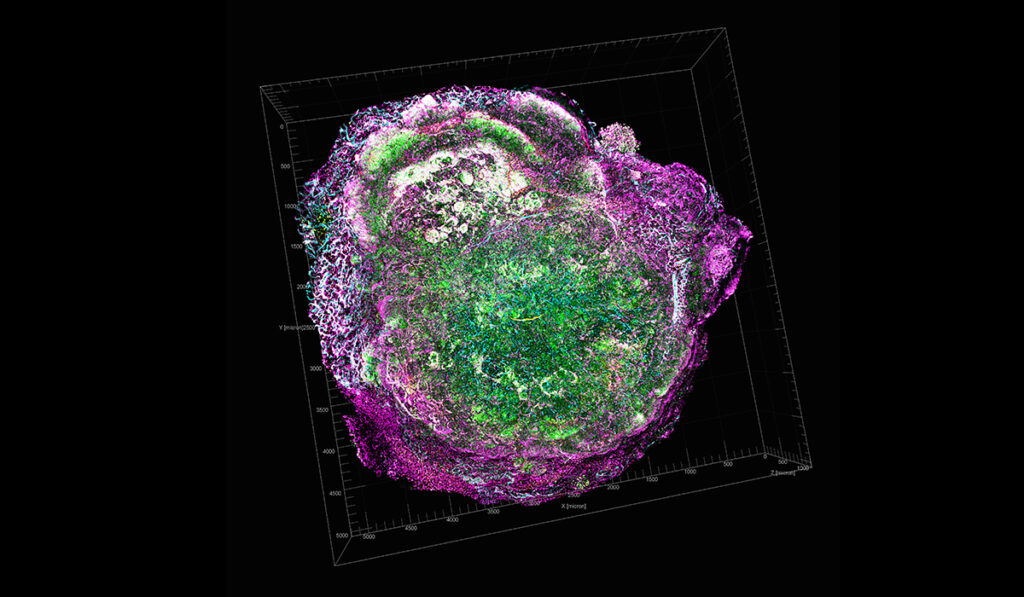
A team of investigators at Vanderbilt University Medical Center has discovered that blocking CXCR2 signaling boosts antitumor immunity and reduces tumor growth and metastasis in models of breast cancer and melanoma.
The findings, reported in the journal Cancer Immunology Research, add mechanistic insight and support for the therapeutic potential of SX-682, a CXCR2 antagonist that is currently being tested in clinical trials for various types of cancer.
MDSCs in the Tumor Microenvironment
One way by which tumors evade the body’s immune response is by secreting chemokines that recruit myeloid-derived suppressor cells (MDSCs). These cells originate in the bone marrow and can suppress the T cell-mediated immune response.
Ann Richmond, Ph.D., Ingram Professor of Cancer Research at Vanderbilt, and her team have long studied tumor-secreted chemokine factors and their receptors on MDSCs, with a particular focus on the receptor CXCR2. CXCR2 is a key chemokine receptor involved in the movement of MDSCs into developing tumors and the pre-metastatic niche.
“We had the hypothesis that if we could target CXCR2 in myeloid cells, we might ablate the trafficking of MDSCs into the tumor microenvironment and thereby enhance the immune response to a developing tumor,” Richmond said.
Stopping MDSC Trafficking
To test the hypothesis, the researchers, led by Jinming Yang, Ph.D., and Chi Yan, Ph.D., of Vanderbilt, developed a mouse model with a targeted deletion of Cxcr2 in myeloid cells and studied the development of tumors from implanted breast cancer or melanoma cells. For both types of implanted tumors, they found reduced tumor growth and metastasis in mice missing Cxcr2 signaling in myeloid cells.
An in-depth analysis of immune cells and chemokine factors in the tumors revealed surprises, Richmond said. In addition to a reduction in MDSCs in the tumors, the researchers found increased numbers of B cells (of the B1b type) and CD8 positive T cells, which kill tumor cells, along with increased levels of the chemokine CXCL11.
“This is a paradigm shift – the idea that by simply blocking CXCR2 expression by myeloid cells, there is an influx of B cells making CXCL11, which then recruits T cells into the tumor microenvironment,” Richmond said.
“By simply blocking CXCR2 expression by myeloid cells, there is an influx of B cells making CXCL11, which then recruits T cells into the tumor microenvironment.”
“Moreover, since the loss of CXCR2 expression in myeloid cells led to depletion of MDSCs, there is also reduction in MDSC suppression of the CD8 positive T cell antitumor activity,” she explained. “The final result is stronger antitumor immunity that suppresses tumor growth and metastasis.”
Mechanistic Insight into SX-682
Importantly, Richmond noted, treatment of tumor-bearing mice with SX-682 to block CXCR2 signaling also inhibited tumor growth, reduced intratumoral MDSCs, and increased intratumoral B1b cells and CD8 positive T cells.
“The CXCR2 antagonist we used, SX-682, is currently in clinical trials in combination with immune therapy for patients with melanoma, and our studies have now substantiated the intricate mechanism that’s involved,” Richmond said.
“Patients don’t all respond to any one therapy, so being able to see how these various immune cells are being modulated with CXCR2 antagonist provides mechanistic insight into how CXCR2 antagonist combined with immune checkpoint inhibitor therapies might work in patients.”
“The final result is stronger antitumor immunity that suppresses tumor growth and metastasis.”
Correlations Found from Clinical Query
To explore whether the B cell-CXCL11 signaling axis may be important in breast cancer and melanoma prognosis, the researchers queried databases of human molecular cancer data (TCGA and GEO-NCBI). They found positive correlations among B cell markers, CXCL11 and a CD8 positive T cell marker in association with increased survival among breast cancer and melanoma patients.
“These data appear to confirm that B cells and CXCL11 are working together to bring in more T cells to enhance tumor killing and survival,” Richmond said.
Moving forward, the researchers are testing combinations of CXCR2 inhibitors, immune therapies and other signaling pathway inhibitors in various cancer models.