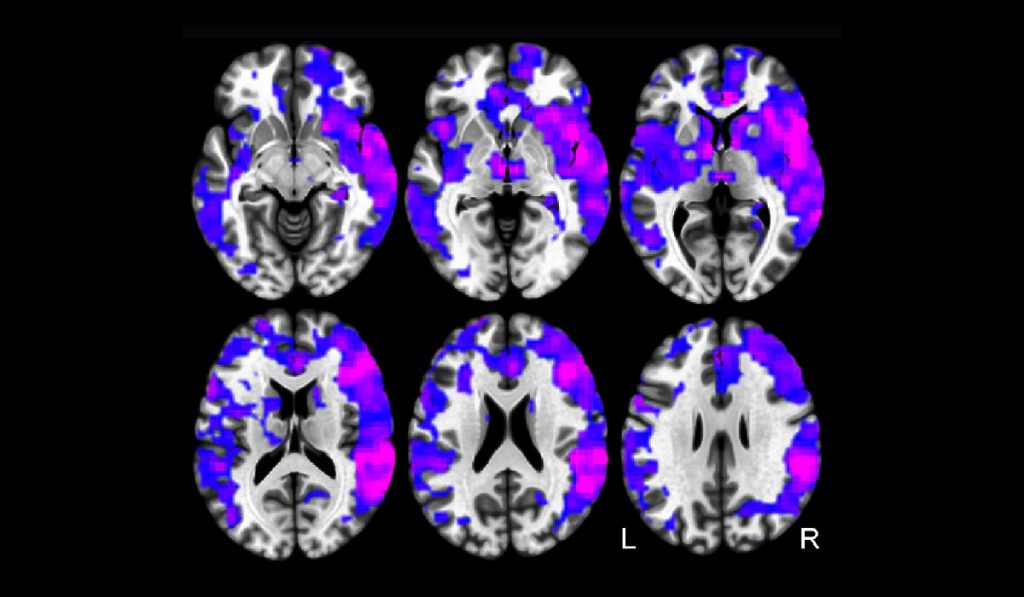
While studies have focused on brain connectivity in epilepsy patients, few have examined brainstem connectivity and its role in epilepsy or neurocognitive disorders. However, many of the subcortical regions most critical for neocortex activation and arousal are contained in the brainstem’s ascending reticular activating system (ARAS).
Reduced ARAS Functional Connectivity in TLE Patients
In a recent study published in the Journal of Neurology, Neurosurgery and Psychiatry, researchers at Vanderbilt University Medical Center used functional magnetic resonance imaging (fMRI) to examine functional connectivity of selected ARAS structures in temporal lobe epilepsy (TLE), the most common cause of focal seizures. The case-control study explored the hypothesis that TLE leads to subcortical arousal center dysfunction, contributing to disturbances in neocortical connectivity and neurocognition.
“We believe the role of brainstem networks has been underappreciated as a contributor to neurocognitive problems,” explained Dario Englot, M.D., surgical director of epilepsy and assistant professor of Neurological Surgery, Radiology and Radiological Sciences, and Biomedical Engineering. “Our objective was to demonstrate how connectivity disturbances in brainstem arousal centers may contribute to cognitive and functional changes associated with TLE.”
Englot, along with Victoria Morgan, Ph.D., associate professor of Radiology and Radiological Sciences, and collaborators from Vanderbilt’s Biomedical Engineering, Psychiatry and Behavioral Sciences and Institute of Imaging Science divisions, examined the cuneiform/subcuneiform nuclei (CSC), pedunculopontine nucleus (PPN), ventral tegmental area (VTA) and other structures in TLE patients. ARAS connectivity patterns were related to neuropsychological and disease measures.
Compared with controls, TLE patients demonstrated reductions in ARAS functional connectivity. Decreased ARAS connectivity was related to increased frequency of seizures and was associated with impairments in verbal IQ, attention, executive function, language and visuospatial memory on neuropsychological evaluation. This and subsequent studies may ultimately lead to novel targets for neuromodulation.
A Model for Predicting TLE Surgery Success
Of patients who have surgery for TLE, only about 60 to 70 percent will remain seizure-free after three years. It is difficult to predict beforehand which patients will or will not benefit from the procedure. In an earlier study, Morgan, Englot and their colleagues computed a model of functional and structural connectivity in the brain associated with presumed TLE seizure propagation using MRI.
The researchers showed that deviance from the model predicted those patients who would experience immediate seizure recurrence after surgery with 100 percent accuracy in a small sample size. This study demonstrated the potential for using connectivity as a clinical tool for identifying the best candidates for epilepsy surgery.
“Current clinical assessments only consider the seizure focus,” said Morgan, “but we now have evidence that previously indistinguishable TLE subtypes with different treatment outcomes can be identified pre-surgically by their long-range networks across the brain.”
Brain Connectivity Post-TLE Surgery
The Vanderbilt investigators are now shifting their focus to brain connectivity following epilepsy surgery. At the recent North American Neuromodulation Society annual meeting, they presented findings from a new study currently underway. Supported by their MRI studies of pre-operative patients with TLE, they discovered that certain impairments in brainstem-neocortical connectivity observed in TLE may recover with successful epilepsy surgery.
“Understanding brain connectivity will help us evaluate the behavioral and cognitive effects of TLE seizures over time,” said Englot, “and will lead to better treatments for preventing the damaging effects of these seizures. If we can address aberrant connectivity patterns, it could have a huge impact on patients’ quality of life.”