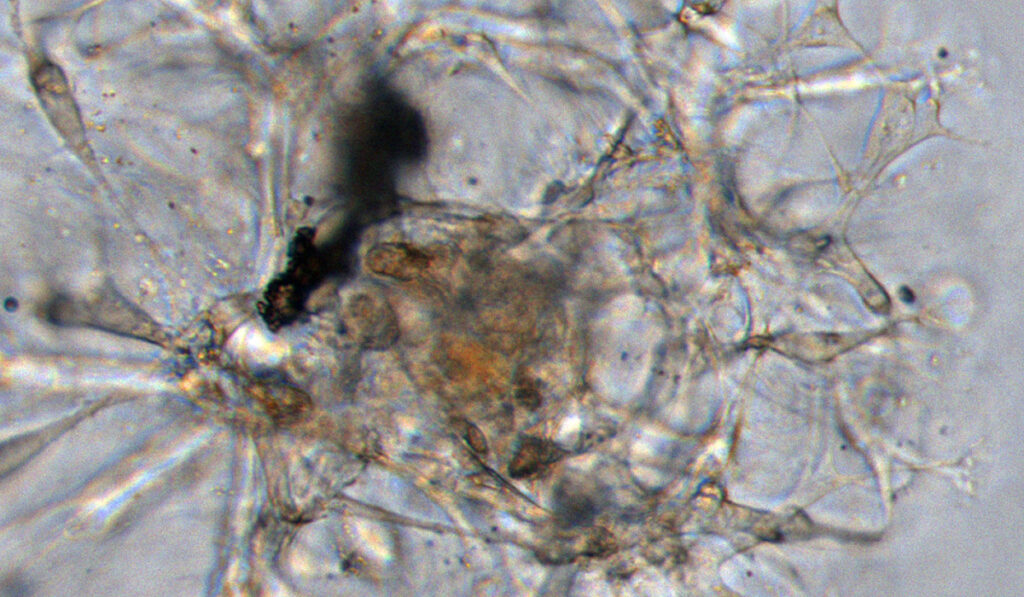
The vast majority of papillary thyroid cancers are slow-growing and typically managed successfully with surgery, with or without radioactive iodine treatment. However, for the subset of these cancers that become aggressive, treatment resistant or widely metastatic, there are very few effective therapies.
This challenge spurred a team of researchers at Vanderbilt University Medical Center to seek molecular targets to cripple the antioxidant defense system supporting the proliferation and growth of metastatic papillary thyroid cancer cells.
Led by Naira Baregamian, M.D., an assistant professor of surgery in the Division of Surgical Oncology and Endocrine Surgery, and Sekhar Raja Konjeti, Ph.D., a staff scientist in the Department of Surgery, the team revealed through a series of in vitro studies the enzyme glutathione peroxidase 4 (GPX4) to be a potential therapeutic target.
GPX4, they found, is more highly expressed in thyroid cancer cells than in normal thyroid tissue and contributes to reduced overall survival. They also report that pharmacologic inhibition of GPX4 with the small molecule RSL3 induces ferroptosis, a form of programmed cell death catalyzed by iron.
“GPX4 also appears to play a major role in other tumor systems,” Baregamian said. “Looking for the key to dismantling this effective, potent antioxidant shield in thyroid cancers is very promising and exciting.”
Vanderbilt surgical resident David Hanna, M.D., and medical student Sriram Cyr also contributed to the work, which won a first place award at the 2021 Cancer Research Paper Competition, an annual event hosted by the American College of Surgeons Commission on Cancer and the American Cancer Society.
“When ferroptosis occurs on a broad scale, then you really are weakening the tumor and there is no way for the cancer cells to come back from it.”
Isolating the Target
Oxidative stress is necessary for the production of thyroid hormone; thus, both healthy and malignant thyroid cells live in a highly oxidative environment. Yet, oxidative stress also drives a high rate of ferroptosis, so the growth and survival of both healthy and malignant cells depends on a sophisticated antioxidant system to regulate this environment and temper ferroptosis.
Through metabolomic studies, Baregamian and her team found that malignant thyroid tissues exhibit significant upregulation of these protective antioxidant mechanisms compared to normal thyroid tissues. They also discovered that the enzyme GPX4 is central to this protective function.
“Antioxidants detoxify chemicals like those used in chemotherapy,” Konjeti said. “As we know from other cancers, when tumors have a high antioxidant makeup, this helps them evade the effect of the treatment. GPX4 counters oxidative stress, so it can render our therapies impotent.”
This unveiled a targeted need to understand how GPX4 alters the tumor microenvironment to potentiate therapeutic resistance in thyroid cancers.
“Importantly, when the cells are blocked from utilizing GPX4 to mitigate toxins, you dismantle the antioxidant capacity of the cell – its main line of defense – and ferroptosis may be unleashed,” Konjeti said.
“When ferroptosis occurs on a broad scale, then you really are weakening the tumor and there is no way for the cancer cells to come back from it,” Baregamian said.
RSL3 as GPX4 Disruptor…and More
The team’s search for an inhibitor led them to the small molecule RSL3. In the preclinical models, this molecule was found to profoundly disrupt the function of GPX4. Even when tested it in doses needed to perform effective inhibition, they found that RSL3 spared healthy cells.
The team found yet more to mine from the RSL3 molecule. The introduction of RSL3 produced not only cell death, but suppression of the mTOR pathway and its downstream signaling proteins.
This is significant because a large percentage of papillary thyroid cancers have BRAF V600E mutations, and these cancers utilize the mTOR pathway to enable the tumor to progress and proliferate. Baregamian says this was a novel finding, as the specific parts of the mTOR pathway that were suppressed by RSL3 are not described in other cancers.
Further, they also found that RSL3 treatment not only led to increased DNA damage, but also incapacitated the DNA damage repair responses within thyroid tumor cells.
Beyond Preclinical Models
Baregamian says next steps are to test RSL3 and similar GPX4-targeting molecules in patient-derived thyroid cancer organoid models and in vivo to gain clear insight.
“We also have a lot of work ahead of us to decipher the impact of GPX4 in therapy-resistant thyroid tumors with diverse mutational signatures,” she said. “Knowing this will allow a precision oncological approach to treating thyroid tumors.”