Uveal coloboma represents one of the most common congenital ocular malformations, accounting for up to 10 percent of childhood blindness. Coloboma originates from defective fusion of the optic fissure, and genetic heterogeneity combined with the activity of developmentally regulated genes suggests multiple mechanisms regulate optic fissure closure.
Mutations in the gene encoding neurofibromin 2 (NF2), a tumor suppressor protein important in development and regeneration in diverse organs and tissues, can cause eye abnormalities in humans, including coloboma. However, the precise role of NF2 in optic fissure closure is not clear.
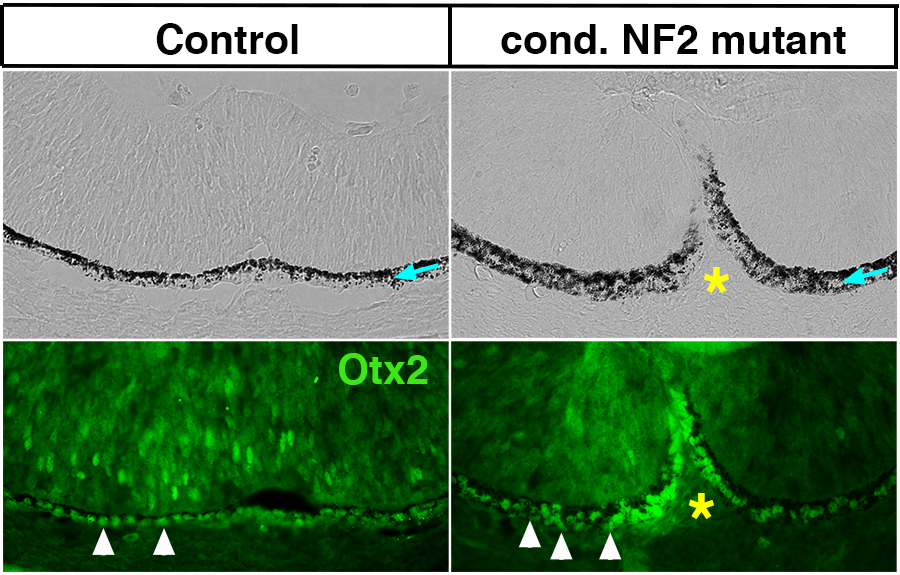
In a study published in Human Molecular Genetics, researchers at the Vanderbilt Eye Institute used genetic strategies to inactivate Nf2 in the developing mouse eye. Their findings indicate that loss of Nf2 function and expansion of the retinal pigmented epithelium (RPE) is a novel underlying cause of coloboma.
“We know that limiting proliferation – particularly in the RPE layer – is a critical mechanism during the closure of the optic fissure,” said Sabine Fuhrmann, Ph.D., an associate professor in the Department of Ophthalmology and Visual Sciences at Vanderbilt University Medical Center. “In general, mutations in several genes cause congenital coloboma but we do not understand how many of these important genes actually regulate the closure process at the cellular level. We learned from our study that ensuring correct production of RPE cells may be essential at a critical time during the closure process.”
“We learned from our study that ensuring correct production of RPE cells may be essential at a critical time during the closure process.”
The Role of NF2
NF2 controls diverse processes in cancer, development, and regeneration via the Hippo pathway and cytoskeleton regulation. In the embryonic eye, NF2 is required for development of the lens and the optic cup, particularly the ciliary body and iris. NF2 mutations cause a variety of ocular defects, particularly in the RPE, including combined retinal and RPE hamartomas.
In the developing optic cup, the RPE slowly proliferates to maintain a single layer and differentiates into pigmented cells. Specifically, embryonic mouse eyes with Nf2 mutations show substantially increased RPE proliferation in the fissure region.
Coloboma occurs in mice upon tissue specific Nf2 deletion; however, it is unclear how loss of Nf2 function affects the closure process. “We know NF2 is required for balancing RPE cell number,” Fuhrmann explained. “Active proliferation in the presumptive RPE layer occurs early in eye development but then has to slow down. That’s not happening in this coloboma mutation.”
It’s possible that the loss of expression of Nf2 results in cellular crowding, preventing cells from re-orienting properly in the optic fissure, she added.
Proliferation of RPE Cells
Using conditional inactivation of Nf2 in an embryonic mouse model, the researchers discovered that eyes lacking Nf2 had increased proliferation of RPE cells in the optic fissure region. The cells failed to transition to a cuboidal shape, resulting in insufficient alignment, failure to fuse and persistent coloboma.
“What’s really exciting to us is that these hyperproliferating RPE cells maintain RPE character.”
Additionally, the researchers found that during hyperproliferation of RPE cells, instead of one single layer the RPEs formed multiple layers. “What’s really exciting to us is that these hyperproliferating RPE cells maintain RPE character,” Fuhrmann said. “Normally, they would be lost by transforming into another cell type – the retina – in the embryonic mouse eye.”
Implications for Regeneration
Understanding the role of Nf2 in RPE proliferation in mice may have relevance for regenerative therapies in adult eyes. Fuhrmann and colleagues are testing other genetic and pharmacological manipulations to prevent the transformation of RPE cells into different cell types.
“When adult eye tissue is damaged under trauma or other circumstances, RPE cells can also start to proliferate but they transform pathologically into different cell types,” Fuhrmann said. “Some of these transformed cells go rogue and migrate into the vitreous contributing in formation of pathological epiretinal membranes.”
“If the RPE can proliferate and produce new RPE cells with this particular genetic manipulation during development, it may help to understand what is normally preventing regeneration of the adult RPE.”