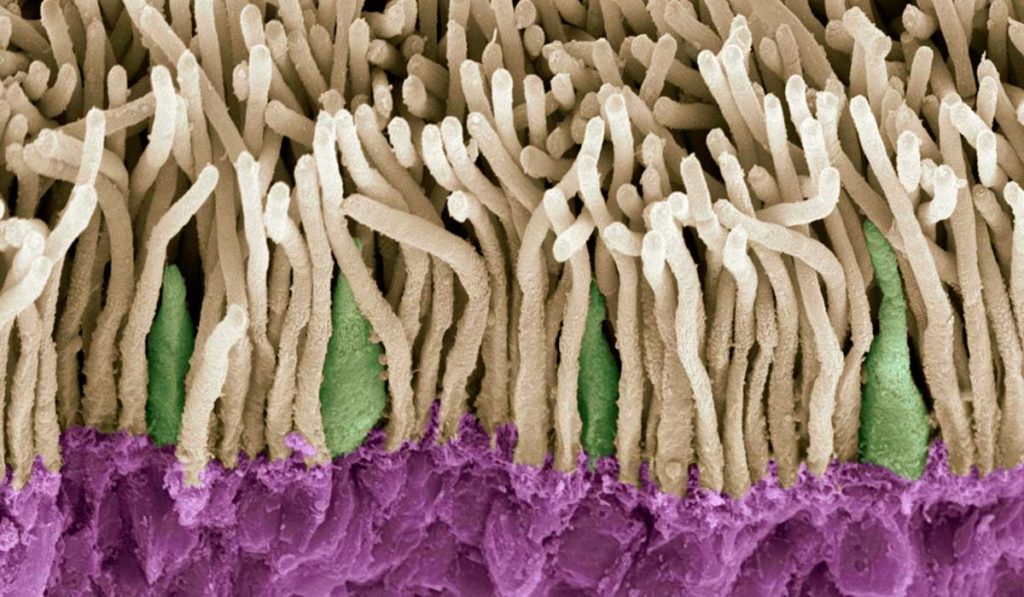
For decades, diabetic retinopathy (DR) has been considered primarily a disease of retinal blood vessels. It has become increasingly clear, however, that diabetes affects not only retinal vasculature, but also retinal neurons like photoreceptors. Retinal neuron dysfunction may precede the earliest manifestations of diabetic retinal vasculopathy, suggesting that retinal neurons may play a role in disease progression.
“Photoreceptors are the most numerous neurons in the retina and have been shown to be the most metabolically active,” said Irina De la Huerta, M.D., assistant professor of ophthalmology at Vanderbilt University Medical Center. “We know that photoreceptors are affected by high blood sugar levels and they may contribute to the development of retinopathy in patients with diabetes. However, the mechanisms by which high glucose affects photoreceptors, and by which photoreceptors under high sugar conditions, in turn, impact the retinal vasculature, are not well understood.”
De la Huerta received a Career Starter Grant from the Knights Templar Eye Foundation and a grant from the International Retinal Research Foundation to investigate how photoreceptors are impacted by elevated glucose levels.
“We are beginning to explore how this new pathway affects the progression of diabetic retinopathy, using cell culture and diabetic animal models.”
Following the Mediators
De la Huerta and colleagues began investigating the effect of high glucose on photoreceptors. By comparing gene expression and protein production by photoreceptors from diabetic versus nondiabetic retinas, they identified pro-inflammatory mediators that are produced by photoreceptors under high glucose conditions.
Some of these mediators – including TNF-α, IL-1β, and IL-1α – are known to be involved in modulating processes leading to DR progression. By following the mediators, they uncovered the pathway involved in the response of photoreceptors to elevated glucose conditions.
“We are beginning to explore how this new pathway affects the progression of diabetic retinopathy, using cell culture and diabetic animal models,” De la Huerta said. “By understanding the mechanisms and the role of this pathway, we hope to identify solutions to prevent the retinopathy from progressing.”
Capturing Photoreceptors
De la Huerta and her research team have also isolated photoreceptors and begun to study them in culture. “Photoreceptors are difficult to study,” she said. “Vascular endothelial cells and glial cells can be observed in culture because they divide, but photoreceptors, like all neurons, don’t. The photoreceptors that you isolate from the retina are what you have to work with.”
The team has developed a system to isolate photoreceptors from all other cells in the retina and establish a photoreceptor culture. Using immunostaining for known photoreceptor markers and microscopy, they first confirm that the cells isolated are photoreceptors and that the culture is pure. They then expose the photoreceptor culture to high glucose levels. RNA sequencing and ELISA protein assays show which genes are expressed and which proteins are produced at a higher level in cells exposed to elevated glucose.
“This work has the potential for significant socioeconomic impact; diabetic retinopathy is the leading cause of blindness in working age adults.”
Moving Forward
De la Huerta was recently selected by the National Association for Eye and Vision Research to present this work as an example of promising vision research on Capitol Hill to support congressional funding for the National Institutes of Health and the National Eye Institute.
“This work has the potential for significant socioeconomic impact; diabetic retinopathy is the leading cause of blindness in working-age adults,” she said. “Next we will be looking at therapeutic interventions that would reduce the deleterious effects of glucose on photoreceptors.”