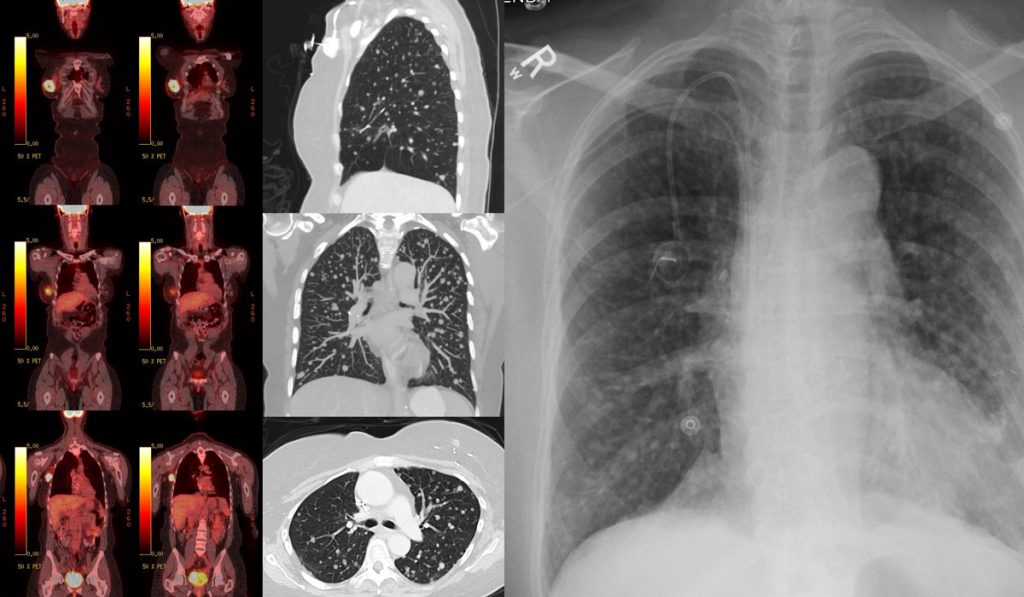
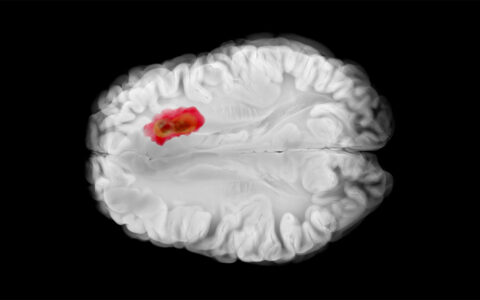
Positron emission tomography (PET) holds unexploited promise for viewing dynamic tumors precisely wherever they “live.” It opens the door to developing precision radiopharmaceuticals (tracers) that can perform more functions and elucidate more types of tumors, in more stages. It may also usher in therapies that target the metabolic vulnerabilities tracers identify.
“Positive emission topography tells us what is on the cancer cell’s ‘mind’ at a given time and location,” said Charles Manning, Ph.D., director of the Vanderbilt Center for Molecular Probes and Molecular Imaging Research. “It gives us the ability to understand the distinguishing features of a patient’s cancer, and because it’s quantitative, we can ask it, ‘how much’?”
“PET is uniquely positioned to identify certain phenotypical features that would be targetable by therapy.”
Manning is a co-principal investigator of a National Cancer Institute-funded initiative, VU-PREDICT, that will develop predictive imaging technology for cancer. “In the field of drug development for cancer, there are not a lot of predictive biomarkers. PET is uniquely positioned to identify certain phenotypical features that would be targetable by therapy,” Manning said.
New Tracers and Therapy Targets
“Dr. Manning and the Vanderbilt Institute of Imaging Science do amazing work and have contributed novel imaging to our clinical trials. This initiative will help us to do even more and to collaborate with colleagues around the country in using imaging to improve precision therapy,” said Jordan Berlin, M.D., associate director of Clinical Investigation Strategy at Vanderbilt-Ingram Cancer Center.
Together, the team is looking at investigational tracers with different mechanisms for elucidating tumor metabolism. “Currently used tracers, like the work horse FDG 18 (which illuminates glucose uptake), light up the tumors in many, many kinds of cancer. But FDG 18 does not do this in pancreatic cancer, for example, nor does it distinguish nodular from cancerous lung lesions,” Manning said. A glutamine uptake tracer, however, may.
“If we can use our experimental tracer to see that the tumor is using glutamine to fuel itself,” Manning said, “this paves the way for targeted therapy.”
PET Drives Research
In one clinical trial, the team is imaging patients with metastatic colorectal cancer. “We want to look at the accumulation of radiotracers targeting metabolism to find the Achilles heel for that tumor,” Manning said. From these data, they plan to investigate several different classes of cancer therapies.
In parallel, they are using patient-derived xenograft mouse models of colorectal cancer to refine PET imaging techniques and test the experimental radiopharmaceuticals. “Colorectal cancer is our initial focus. But in principle, many of the things we are working on are applicable to other solid tumors. We anticipate going beyond colon cancer and looking at melanoma, breast cancer and lung cancer as well,” Manning said.
Refining Treatment Regimens
The studies may bring cancer therapy closer to targeted treatment. “Currently, we may know that a certain drug tends to work well on a certain mutation of a certain type of cancer in situ. If that drug doesn’t work, the second-line therapy is introduced,” Manning explained. Manning anticipates clinicians evaluating patients’ tumors with PET very soon after the first round of therapy to try to predict their response. “Doing these tests early, if the patient is not responding, we can move them to something more likely to work,” he said.
Sequential PET scanning with multiple tracer tools may be particular important with metastases because a drug that worked for the primary tumor can make a metastatic tumor worse.
Manning also envisions using tracers that double as therapy, a concept dubbed “theranostics.” Imaging is done with the tracer and if there is accumulation, the clinician matches that area with an infusion of the therapeutic drug in a sequential, same-day procedure. This has been highly successful in using Lutetium PSMA for prostate cancer.
“We are in a very exciting time right now in this field because the theranostic concept has been embraced by Pharma,” said Manning “We are only scratching the surface of the imaging and therapy partnerships we can develop.”